Recent PROTECT Study uses Computational Modeling of Bubbles to improve electrochemical removal of chemical pollutants in water
A recent Project 4 study reveals efficient ways of exploiting the generated hydrogen and oxygen bubbles by electrochemical methods to enhance removal of contaminants from water. Access to clean water is a global challenge. One method for cleaning contaminated water is applying a low level electric current through electrodes (e.g. an electrical conductor) to generate oxygen and induce oxidation and degradation of organic pollutants, which is the primary focus of PROTECT Project 4.
During the process of cleaning water using electrochemical process, hydrogen and oxygen bubbles are generated as part of electrolysis at the electrodes. Once these bubbles become large enough to detach from the electrode, they start flowing inside the electrochemical cell. As oxygen bubbles continue to form, they will react with the solution to forms oxidants. Our study provides a fundamental understanding on the bubble growth aspect as they develop, dissolve and move in the solution.
“We carried out our study of bubbles growth using computational fluid dynamic techniques in which we numerically solved set of complex equations to describe the fluid dynamics and chemical transport between two phases (e.g. gas bubbles and bulk liquid phases) using computers,” says lead author and former PROTECT Trainee Amir Taqieddin. “We used an advance computational method which couples two numerical approaches (e.g. level set and volume of fluid methods) to describe the dynamics and transport of chemicals across the bubble’s interface from the liquid phase (e.g. water). This advance technique allows us to accurately solve our targeted equations by obtaining sharp bubble interface and therefore predict the interfacial transport between the two phases with high fidelity.”
Understanding the generation, growth, and dynamics of bubbles as they absorb or release dissolved gas in reactive flows is crucial for optimizing the efficiency of electrochemically gas-evolving systems. This will allow development of more efficient electrochemical cells toward cleaning contaminated water. The approach enables the incorporation of electric fields and chemical reactions that are essential for studying the physicochemical hydrodynamics in multiphysics systems. “As PROTECT aims to ultimately provide clean water via electrochemical remediation, we hope that our publication will help PROTECT to better design the targeted remediation cells,” concludes Amir. Amir graduated with MS in mechanical engineering from Northeastern in 2018 under the supervision of Akram Alshawabkeh and Michael Allshouse and is now pursuing his PhD at the University of Illinois Urbana-Champaign.
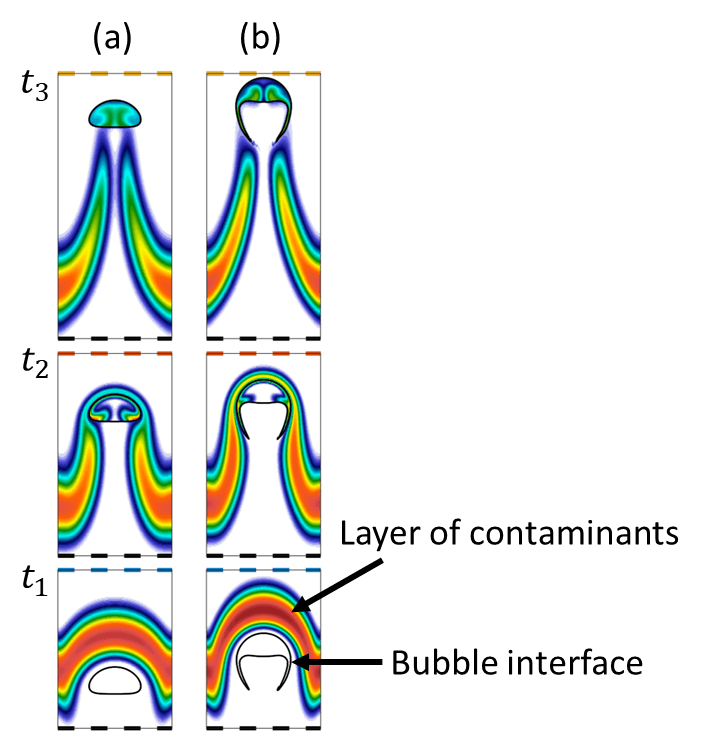
The figure shows the bubble rises inside a liquid domain over time (t_3>t_2>t_1). At time t_1, the bubble reaches a layer of concentrated contaminants in the liquid. We see there are no contaminants inside the bubble at this stage (e.g. the bubble is white from inside).
As the bubble rises and passes through the concentrated layer of contaminants, it absorbs contaminants over time and results a wake behind that drag the concentrated chemicals. In the figure, we studied two types of bubbles based on its shape which are the (a) spherical and (b) skirted bubbles. The bubble shape is controlled by several physical parameters such as the bubble radius, and density and viscosity ratio between the liquid and bubble phases.
